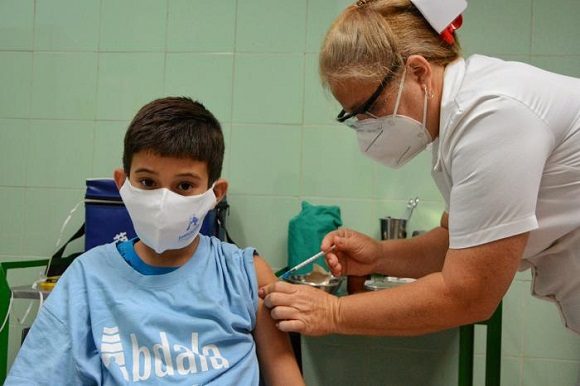
Abdala showed high levels of seroconversion (emergence of antibodies against an infectious disease) in minors between the ages 3 and 18 years, participants in the Ismalall clinical trial.
The Center for Genetic Engineering and Biotechnology (CIGB) reported on its Twitter account that in that study, conducted at the Central Province of Camagüey, volunteers up to 11 recorded an increase of 99.15 percent, while those of 12 At 18, they reported an increase of 98.28 percent.
The Center for Genetic Engineering and Biotechnology (CIGB) reported on its Twitter account that in that study, conducted at the Central Province of Camagüey, volunteers up to 11 recorded an increase of 99.15 percent, while those of 12 At 18, they reported an increase of 98.28 percent.
He specified the publication that the increase in antibody titers was four times or more from the first dose of the drug.

The clinical trial concluded on October 14, 500 children participated and two formulations were used with 25 and 50 micrograms of Abdala, in a scheme of 0-14-28 days.
The #Bdala vaccine showed high levels of seroconversion after vaccination in the strata of ages studied, between 3 and 18 years old during the pediatric clinical studio # Ismalall. # Masqueunavacunaesunpais # Unpawarmorlavida # Cubavive pic.twitter.com/ZX5JS3NUE
The main investigator of studies with that vaccine, Sonia Resik, stood out at that moment that more than 80 percent of adverse events reported in small were mild and the rest of the indicators were comparable to the data obtained in adults, a fact that He ratified his effectiveness.

Abdala earn from July the authorization for use in an emergency, after demonstrating an effectiveness of 92.28 percent in the prevention of the symptomatic disease of Covid-19, and became the first immunogen of Cuba and Latin America.
He also tested an effectiveness of 90 percent in serious patients affected by the Coronavirus SARS-COV-2, causing that condition.
CUBADEBATE
